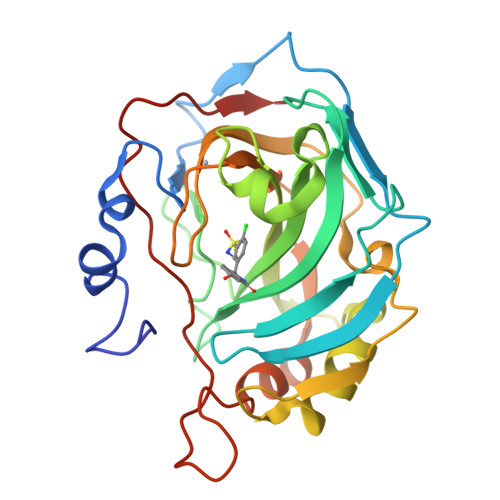
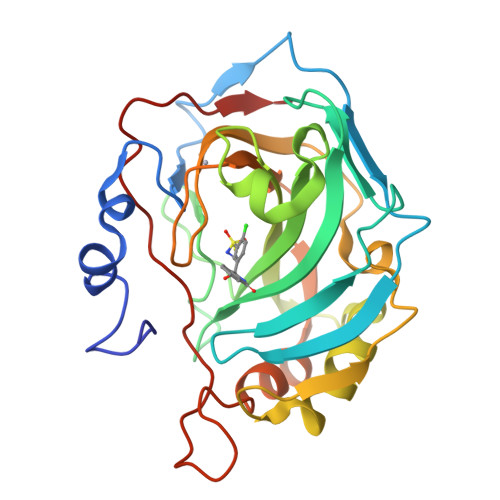
Carbonic anhydrase inhibitors. Comparison of chlorthalidone and indapamide X-ray crystal structures in adducts with isozyme II: when three water molecules and the keto-enol tautomerism make the difference.
Temperini, C., Cecchi, A., Scozzafava, A., Supuran, C.T.(2009) J Med Chem 52: 322-328
- PubMed: 19115843
- DOI: https://doi.org/10.1021/jm801386n
- Primary Citation of Related Structures:
3F4X - PubMed Abstract:
Thiazide diuretics inhibit all mammalian isoforms of carbonic anhydrase (CA, EC 4.2.1.1) with a different profile as compared to classical inhibitors. Acting as moderate-weak inhibitors of CA II and CA I, chlorthalidone and indapamide considerably inhibit other isozymes among the 16 CAs present in vertebrates. These compounds show a different behavior against CAs I and II, with chlorthalidone being 18.3 times more potent against CA II and 150 times more potent against CA I, as compared to indapamide. In the X-ray crystal structures of the CA II-chlorthalidone adduct three active site water molecules interacting with the inhibitor scaffold were observed that lack in the corresponding indapamide adduct. Chlorthalidone bound within the active site is in an enolic tautomeric form, with the OH moiety participating in two strong hydrogen bonds with Asn67 and a water molecule. This binding mode may be exploited for designing better CA II inhibitors.
Organizational Affiliation:
Laboratorio di Chimica Bioinorganica, Universita degli Studi di Firenze, Rm. 188, Via della Lastruccia 3, I-50019 Sesto Fiorentino, Firenze, Italy.